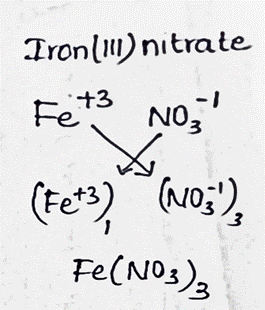
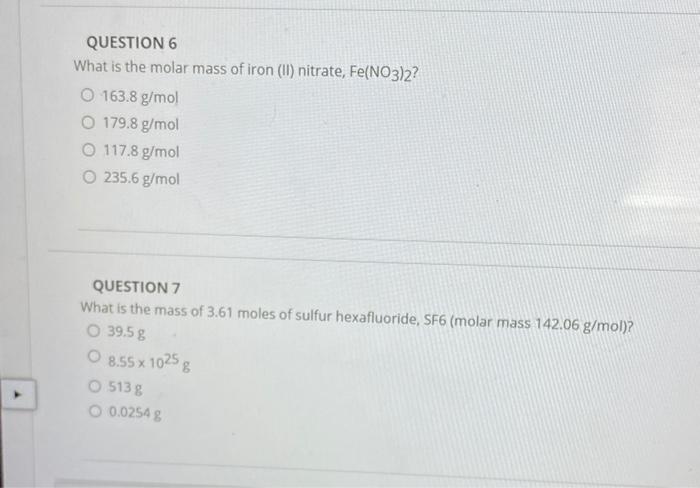
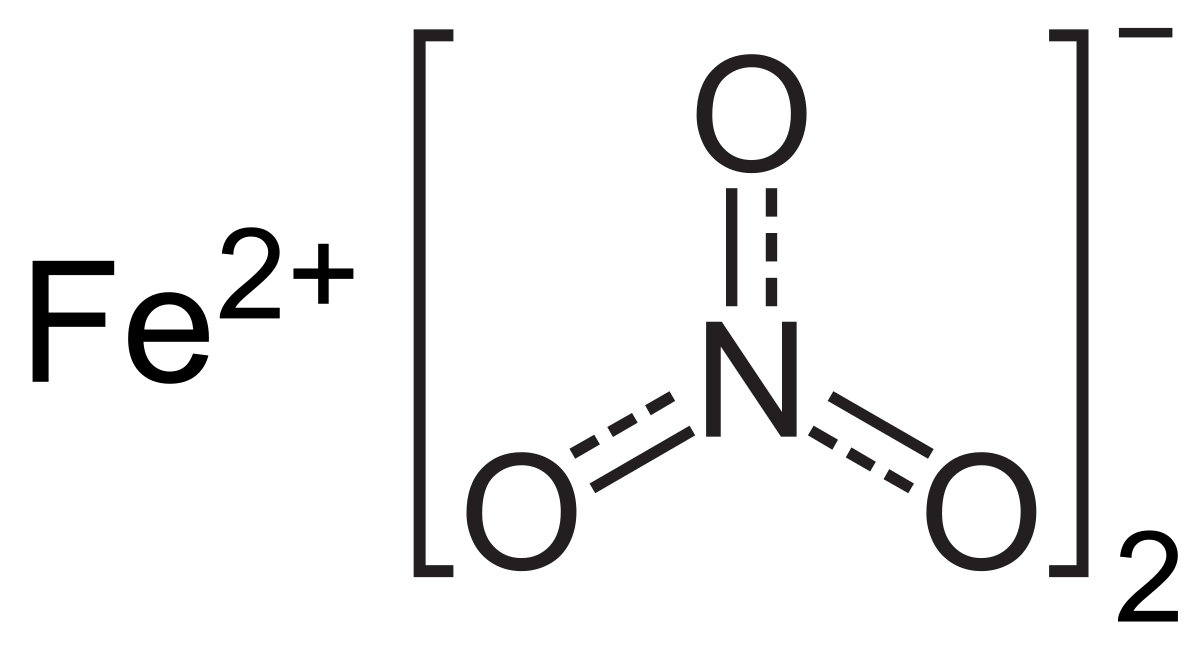
SOLVED:(a) What is the molar mass of iron(II) nitrate, Fe(NO3)2 ? (b) What is the mass, in grams, of 0.200 mol Fe(NO3)2 ? (c) How many moles of Fe(NO3) 2 are present in
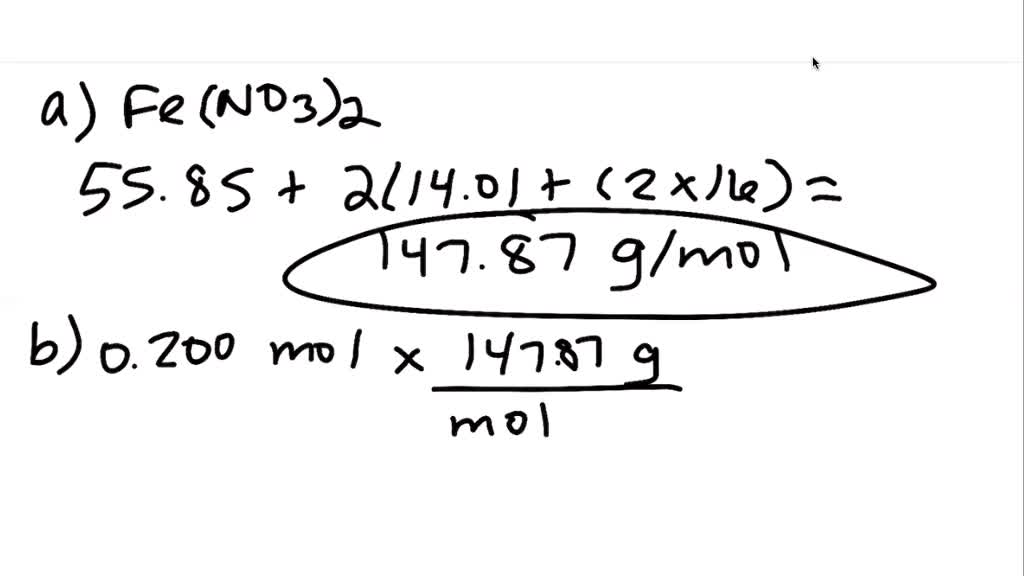
SOLVED:(a) What is the molar mass of iron(II) nitrate, Fe(NO3)2 ? (b) What is the mass, in grams, of 0.200 mol Fe(NO3)2 ? (c) How many moles of Fe(NO3) 2 are present in

SOLVED: What mass of iron(III) hydroxide precipitate can be produced by reacting 75.0 mL of 0.105 M iron(III) nitrate with 125 mL of 0.150 M sodium hydroxide?
WS 7.0 Part II. Molar masses! 1a. The average mass of an iron atom is 9.277 x 10-23 grams (this was discovered by experiments cu

MOLE (mol) Particles (atoms or molecules) DO NOW: Which conversion factor allows you to convert from moles to particles? 1 mol x particles. - ppt download